VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00027
102579-59-9
C₁₃H₁₇N₅O₅S
355.369
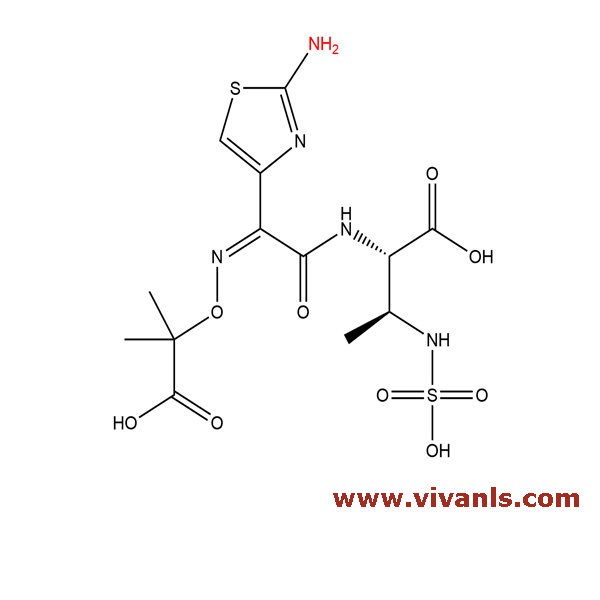
VLIM-00028
87500-74-1
C₁₃H₁₉N₅O₆S
373.384

VLIM-00029
864544-37-6
C₁₈H₂₉NO₅
339.432

VLIM-00030
15258-73-8
C₂₆H₄₀N₂O₅
460.61

VLIM-00031
175481-37-5
C₁₃H₁₈N₂O₃
250.3

VLIM-00032
14943-53-4
C₂₂H₂₅NO₃. HCl
387.19
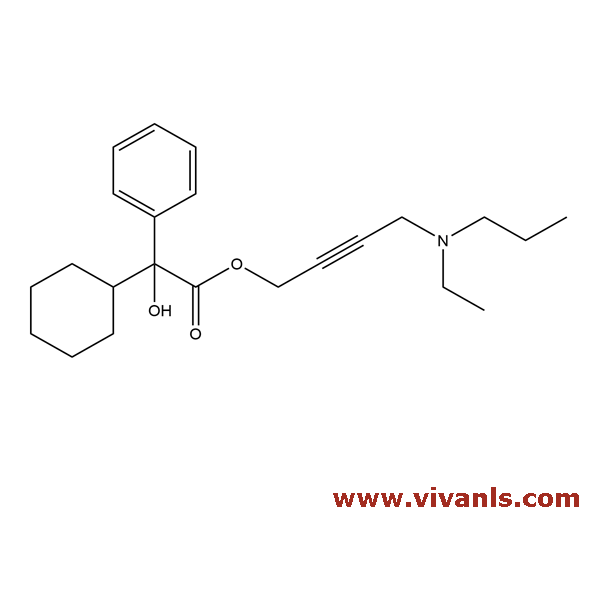
VLIM-00033
1215677-72-7
C₂₃H₃₃NO₃ . HCl
407.19

VLIM-00034
260435-42-5
C₁₃H₁₂N₄O₂
256

VLIM-00035
479035-75-1
C₃₂H₃₉NO₄
501.65

VLIM-00036
192876-02-1
C₁₄H₁₉F₃N₂O₂
304.31

VLIM-00037
177270-91-6
C₁₃H₁₂N₄O
256.26

VLIM-00038
832747-55-4
C₂₃H₂₇FN₄O₃
426.21