VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00052
22431-46-5
C₁₈H₃₃ClN₂O₆S
440.98

VLIM-00053
88699-84-7
C₁₃H₁₆F₃NO₂
275.27
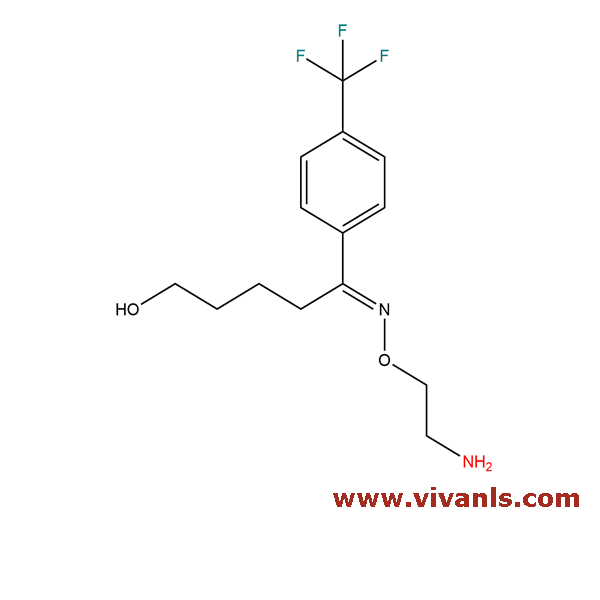
VLIM-00054
192876-02-1
C₁₄H₁₉F₃N₂O₂
304.31

VLIM-00055
220438-80-2
C₁₁H₁₂O₅
224.21

VLIM-00056
99469-99-5
C₁₃H₁₆O₅
252.26

VLIM-00057
219921-94-5
C₂₃H₃₇N₃O₅
435.56

VLIM-00058
147770-06-7
C₂₉H₄₀N₂O₄
480.64

VLIM-00059
63547-24-0
C₁₅H₁₄O₃S
274.33

VLIM-00060
63547-22-8
C₁₅H₁₄O₂S
258.34
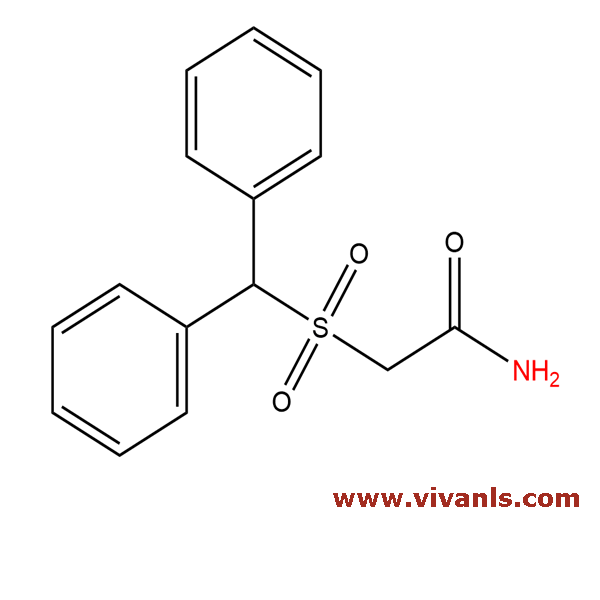
VLIM-00061
118779-53-6
C₁₅H₁₅NO₃S
289.35
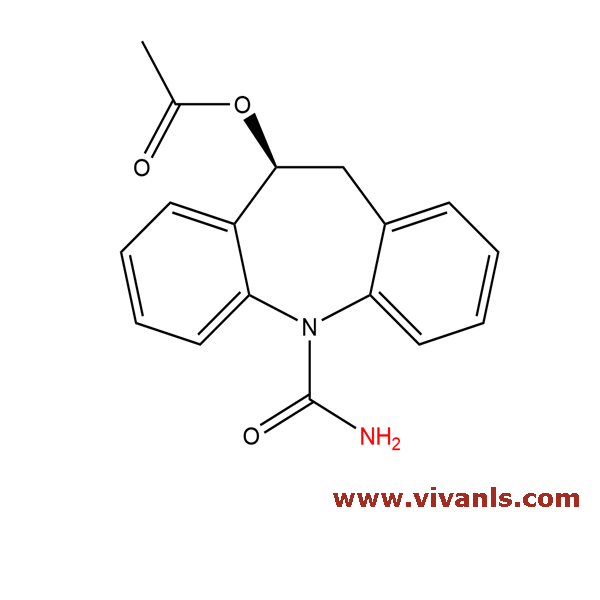
VLIM-00062
236395-14-5
C₁₅H₁₂N₂O
236.27
-1664175477.png)
VLIM-00063
112827-99-3
C₂₇H₄₀O₃
412.605