VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00310
127182-44-9
C₃₉H₇₂N₂O₁₃
776.99

VLIM-00311
691007-09-7
C₂₃H₂₈F₂N₄O₂
430.49

VLIM-00312
1094100-06-7 (acid)
C₂₂H₂₇FN₃O₆S
480.53

VLIM-00314
1262133-64-1
C₂₀H₃₀FN₃O₉
475.46

VLIM-00315
NA
C₂₂H₃₀N₆O₂S
442.58

VLIM-00316
817204-32-3
C₃₁H₂₆FN₃O₇
571.55

VLIM-00317
951380-42-0
C₂₀H₂₁BrFNO₃S
454.35
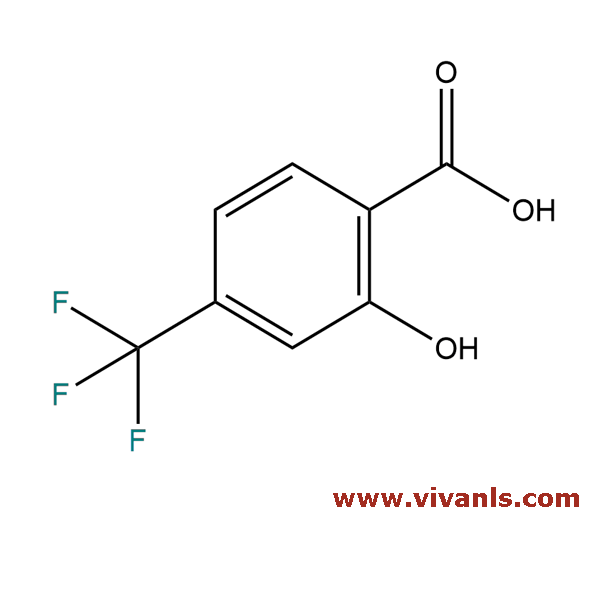
VLIM-00319
328-90-5
C₈H₅F₃O₃
206.12
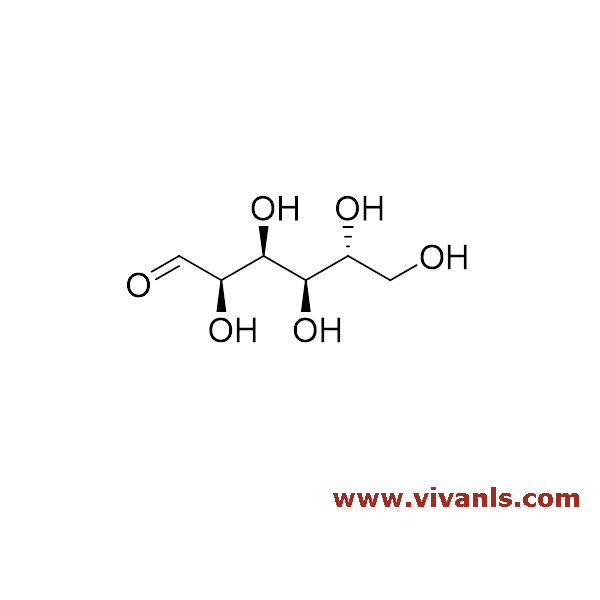
-6-(acetoxymethyl) tetrahydro-2H-pyran-2,3,4,5-tetrayl tetraacetate-1664187129.png)
VLIM-00321
604-68-2
C₁₆H₂₂O₁₁
390.34
-2-(acetoxymethyl)-6-bromotetra hydro-2H-pyran-3,4,5-triyl triacetate-1664187148.png)
VLIM-00322
572-09-8
C₁₄H₁₉BrO₉
411.2
-2-(acetoxymethyl)-3,4-dihydro-2H-pyran-3,4-diyl diacetate-1664187176.png)
VLIM-00323
2873-29-2
C₁₂H₁₆O₇
272.25