VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.
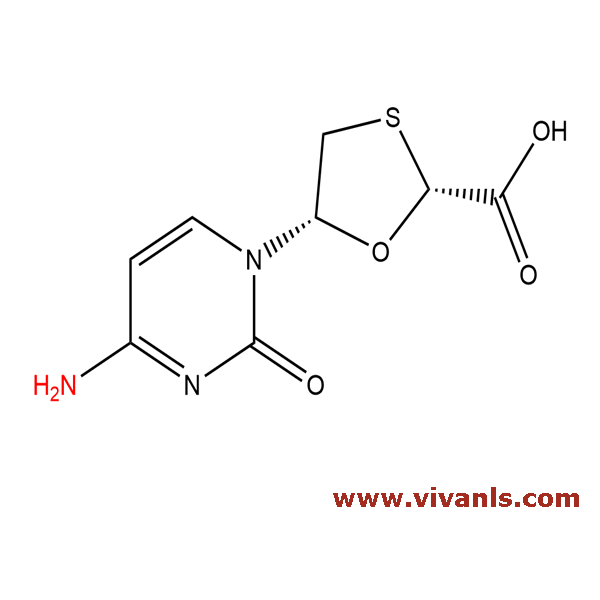
VLIM-00286
173829-09-9
C₈H₉N₃O₄S
243.24
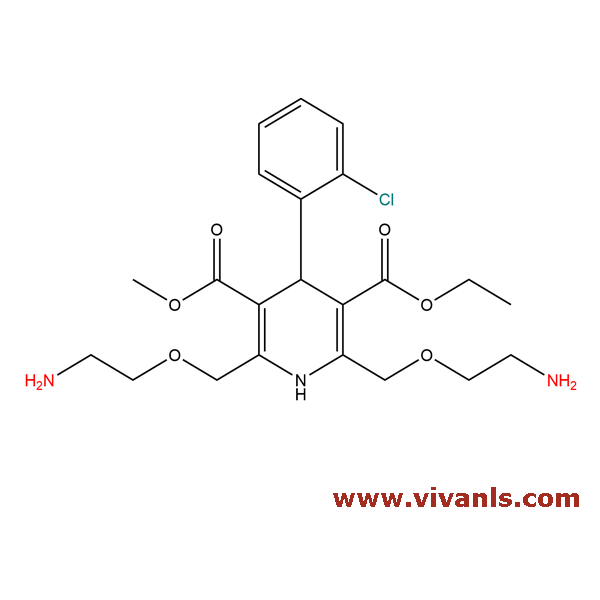
VLIM-00287
721958-74-3
C₂₂H₃₀ClN₃O₆
467.94
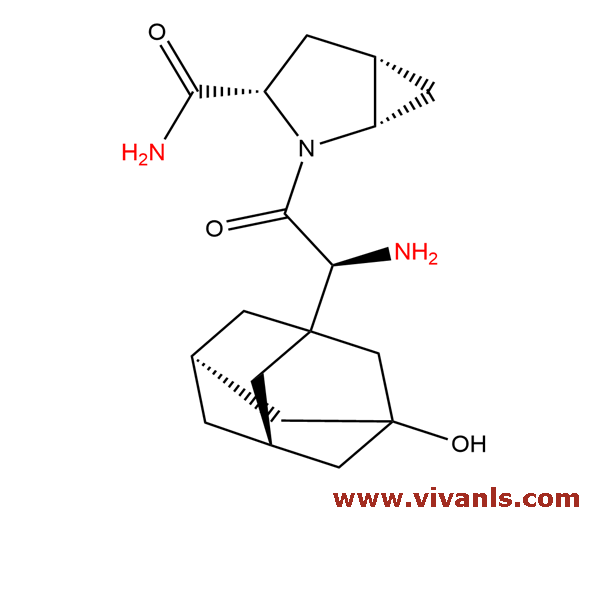
VLIM-00288
1496712-39-0
C₁₈H₂₃N₃O₂
313.4
-1664259335.png)
VLIM-00289
140171-65-9
C₂₁H₂₇ClN₂O₅
422.9

VLIM-00290
252186-78-0
C₉H₆Cl₂N₄O
257.08
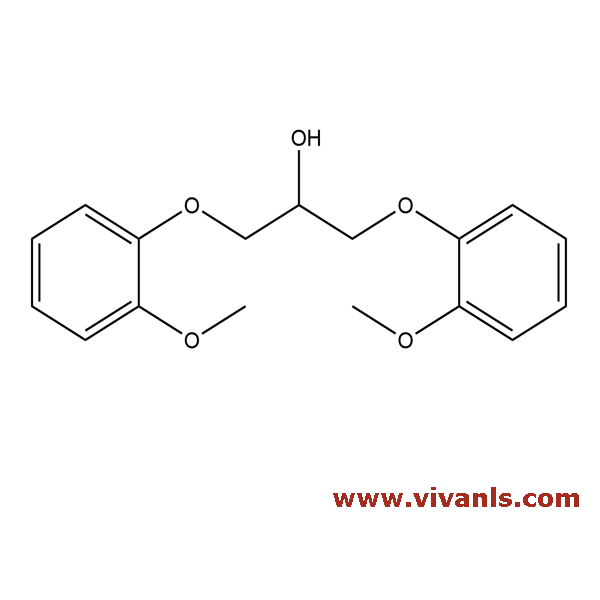
VLIM-00291
16929-60-5
C₁₇H₂₀O₅
304.34

VLIM-00292
1346602-28-5
C₂₄H₂₉FN₄O₂
424.51

VLIM-00293
106266-09-5
C₂₃H₂₈N₄O₂
392.49

VLIM-00294
158697-67-7
C₂₃H₂₇F₂N₃O₂
415.48

VLIM-00295
132961-05-8
C₂₃H₂₈F₂N₄O₂
430.49
-Enalapril Maleate)-1664185806.png)
VLIM-00296
76420-74-1
C₂₀H₂₈N₂O₅. C₄H₄O₄
376.45 116.07
 ethanol-1664185854.png)
VLIM-00297
5197-62-6
C₄H₉ClO₂
124.57