VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00250
23551-25-9
C₂₅H₃₁NO₃
393.52

VLIM-00251
96027-74-6
C₂₃H₂₆ClI₂NO₃
653.72
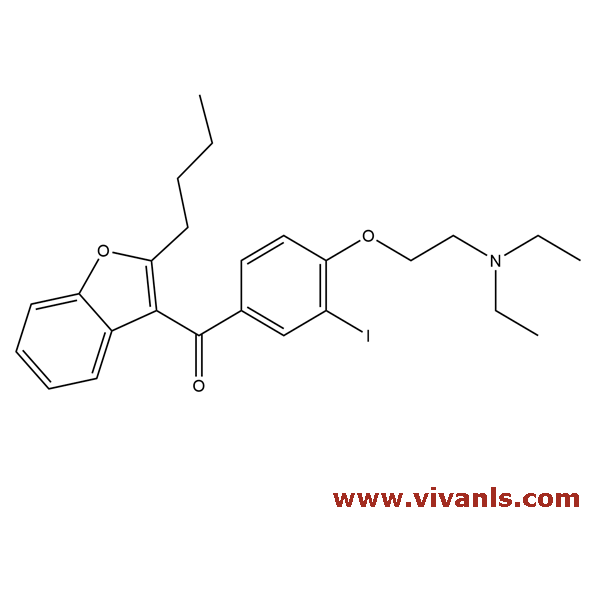
VLIM-00252
85642-08-06
C₂₅H₃₀INO₃
519.42

VLIM-00253
147030-50-0
C₁₉H₁₇IO₃
420.24

VLIM-00254
1087223-70-8
C₂₆H₃₁I₂NO₄
675.34

VLIM-00255
106-47-8
C₆H₆ClN
127.57

VLIM-00256
13951-70-7
C₂₁H₂₈O₆
376.44
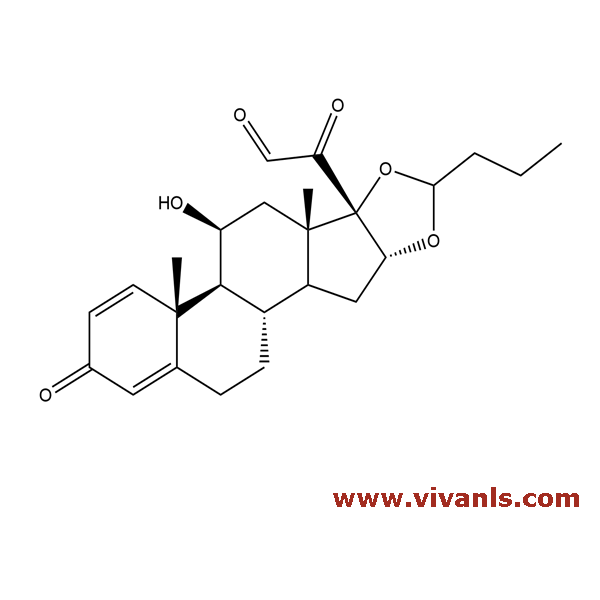
VLIM-00257
85234-63-5
C₂₅H₃₂O₆
428.52

VLIM-00258
638-94-8
C₂₄H₃₂O₆
416.51
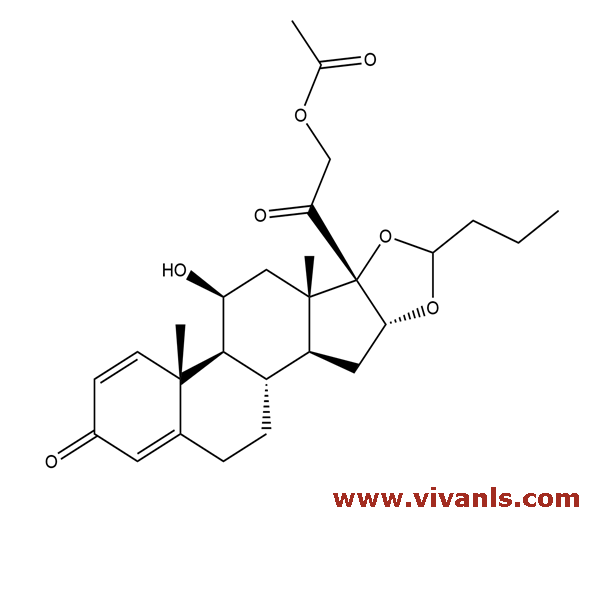
VLIM-00259
51333-05-02
C₂₇H₃₆O₇
472.57

VLIM-00260
70-55-3
C₇H₉NO₂S
171.22

VLIM-00261
27244-64-0
C₉H₁₁NO₅
213.19