VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.
-3-nitrobenzoic acid-1664183919.png)
VLIM-00237
116965-16-3
C₈H₄F₃NO₄
235.12

VLIM-00238
1316291-19-6
C₃₃H₃₄FN₂NaO₇
612.62

VLIM-00239
5452-87-9
C₁₅H₁₉N₃
241.33

VLIM-00240
114248-23-6
C₉H₁₀F₂N₂O₅
264.18

VLIM-00241
87-62-7
C₈H₁₂ClN
157.64
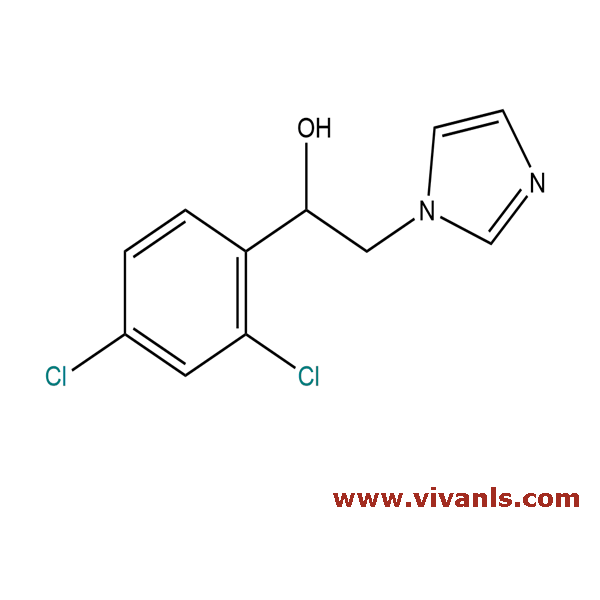
VLIM-00242
24155-42-8
C₁₁H₁₀Cl₂N₂O
256.017

VLIM-00243
913837-72-6
C₁₈H₁₅Cl₃N₂O . HNO₃
381.69
-1664184171.png)
VLIM-00244
52-21-1
C₂₃H₃₀O₆
402.48

VLIM-00245
947502-66-1
C₁₈H₁₈FNO₃S
347.4

VLIM-00246
1391054-37-7
C₁₁H₉FO₂
192.18

VLIM-00248
66774-02-05
C₁₉H₁₅ClO
294.081

VLIM-00249
163222-32-0
C₃₁H₂₇F₂NO₃
499.6