VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00262
60-18-4
C₉H₁₁NO₃
181.19

VLIM-00263
1443331-78-9
C₂₁H₁₆F₄N₄O₃
448.37

VLIM-00264
14007-09-01
C₁₀H₁₄O₄
198.22
![Impurities-7 [5-amino-4-(4-amino-1,3-dioxoisoindolin-2-yl)-5-oxopentanoic acid]-1664260374.png](/images/productimages/Impurities-7 [5-amino-4-(4-amino-1,3-dioxoisoindolin-2-yl)-5-oxopentanoic acid]-1664260374.png)
VLIM-00265
874760-72-2
C₁₃H₁₅N₃O₄
277.28

VLIM-00266
6893-02-03
C₁₅H₁₂I₃NO₄
650.97

VLIM-00267
1131-01-07
C₁₀H₁₂ClNO
197.66

VLIM-00268
120511-72-0
C₂₆H₃₁NO₁₀
517.53

VLIM-00269
103152-84-7
C₁₈H₂₆ClN₃O₄S
415.93
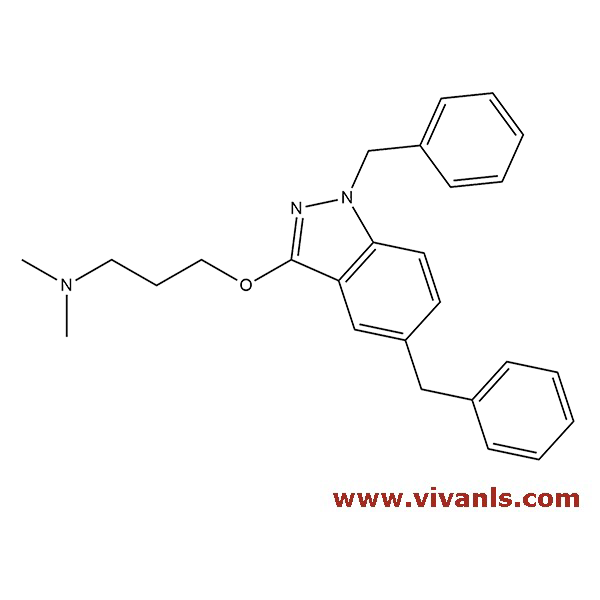
VLIM-00270
1797879-37-8
C₂₆H₂₉N₃O
399.54

VLIM-00271
16391-75-6
C₃₁H₄₈O₆C₄H₁₁NO₂
516.711

VLIM-00272
128486-89-5
C₁₇H₁₈N₄O₂
310.35

VLIM-00273
2215-63-6
C₁₄H₁₂N₂O
224.26