VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.
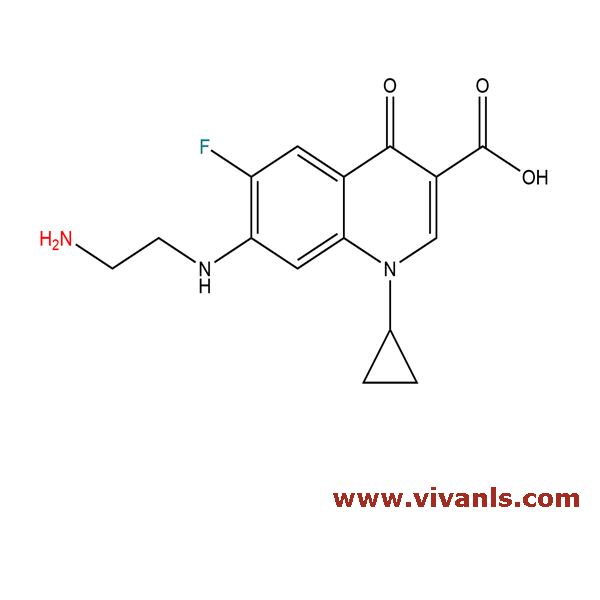
VLIM-00274
103222-12-4
C₁₅H₁₇ClFN₃O₃
341.77

VLIM-00275
66067-44-5
C₁₅H₁₂O₂
224.26

VLIM-00276
140171-66-0
C₁₉H₂₃ClN₂O₅
394.85

VLIM-00277
113994-41-5 (free base)
C₂₀H₂₃ClN₂O₅
406.86

VLIM-00278
43067-01-02
C₁₇H₁₈ClNO₄
335.78
-1664259824.png)
VLIM-00279
93848-82-9
C₂₂H₂₇ClN₂O₆
450.92
-1664259875.png)
VLIM-00280
120289-11-4 (free Acid)
C₁₉H₁₈ClNO₇. 2Na
407.81 2 22.99

VLIM-00281
NA
C₂₇H₂₅ClN₂O₇
524.95

VLIM-00282
34148-67-9
C₁₃H₁₃NO₄
247.25

VLIM-00283
NA
C₂₆H₂₉ClN₂O₇S
549.04

VLIM-00284
3891-07-04
C₁₀H₉NO₃
191.19

VLIM-00285
400602-35-9
C₂₄H₂₉ClN₂O₉
524.96