VIVAN Life Sciences Offers wide range of Impurities / Pharmaceutical Drug Impurities. Impurities are unwanted chemicals that remain with the active pharmaceutical ingredients (APIs) or develop during formulation or upon aging of both API and formulation. The presence of these unwanted chemicals even in trace amount may influence the efficacy and safety of pharmaceutical product. The control of impurities is an important task pharmaceutical impurities as per the regulatory norms. High Pure and Well characterized impurity Standards are used for Related Substances, Organic impurities and Validation of Analytical Methods.
All the Impurity standards are provided with CoA, MASS, NMR & HPLC reports, conforms to Global quality standards.

VLIM-00298
1659301-59-3
C₂₂H₂₅FN₃NaO₅S
485.5

VLIM-00299
837-52-5
C₁₃H₁₄ClN₃
247.73
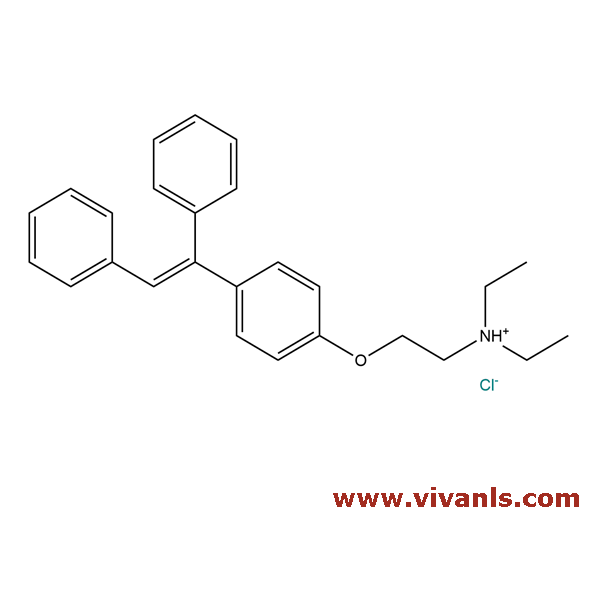
VLIM-00300
144750-42-5
C₁₅H₁₄ClNO₂S · HCl
344.26

VLIM-00301
560075-65-2
C₁₄H₁₀Cl₃NO
314.59

VLIM-00302
748812-53-5
C₂₅H₃₀N₆O₂
446.54

VLIM-00303
4121-40-8
C₈H₉N₅
175.19

VLIM-00304
81886-51-3
C₁₃H₁₃FN₆O
288.28

VLIM-00305
117693-41-1
C₃₀H₅₈N₂O₉
590.79

VLIM-00306
160552-54-5
C₈H₁₁N₃O₄S
245.26

VLIM-00307
144750-52-7
C₁₆H₁₇Cl₂NO₂S
358.28

VLIM-00308
127182-43-8
C₃₇H₆₈N₂O₁₃
748.94

VLIM-00309
127253-06-9
C₃₈H₇₀N₂O₁₃
762.96